|
Georgian Research Institute of Hematology and Blood Transfusion, Tbilisi, USSR. The successes in studying the karyotype of malignant tumors as
well as the achievements in molecular biology and molecular genetics
have considerably deepened our knowledge concerning the role of
chromosomal disorders in oncogenesis [1-4]. Recognition of clonal
origin of leukemias [ 5] has stimulated the interest in studying
the formation and development of cytogenetically marked leukemic
clones during development of leukemias. The purpose of this investigation
was to analyze the pathogenetical and clinical significance of karyotypic
peculiarities in different stages of the leukemic process. Chromosomal
analysis emplyoing the G-banding technique was carried out in 394
patients with preleukemia (PL), secondary leukemia, different stages
of chronic myeloid leukemia (CML), and different variants of leukemia
(AL). The group with PL consisted of 95 patients with firm cytopenia
and either hypo-, normo- or hypercellular bone marrow with erythropoietic
disorders. Cytogenetic examinations revealed only normal diploid
metaphases, but increased percentages of aneuploid and polyploid
cells and structural aberrations, i.e., instability of the karyotype
and clonal anomalies. Karyotypic instability manifested both in
quantitative and structural aberrations. Quantitative disorders
were common for patients with bone marrow hypoplasia, whereas structural
aberrations were more often observed during erythropoietic disorders
(Table 1). Both chromosomal and chromatid types of aberrations were
found, including breaks, fragments, deletions, chromatid exchanges,
marked chromosomes, and, sometimes plural aberrations. We observed
transformation into acute leukemia in 13 out of 88 patients ( 14.8%
). It was found that in patients with karyotypic instability, the
frequency of subsequent emergence of abnormal clones and leukemic
processes is reliably higher (36.4% ) than in patients with normal
karyotype ( 1.8% ). These data suggest that karyotypic instability
may provide a favorable background, or even be the first step, for
the formation of leukemic clones. The dis- . covery of abnormal
clones may be interpreted as evidence of the leukemic process, even
without clinical manifestations. During tumor progression, we observed
an increase in the percentage of abnorma] clones cells as well as
the appearance of new clones. The majority of patients with leukemia
that developed after preceding preleukemic disorders had clonal
abnormalities. 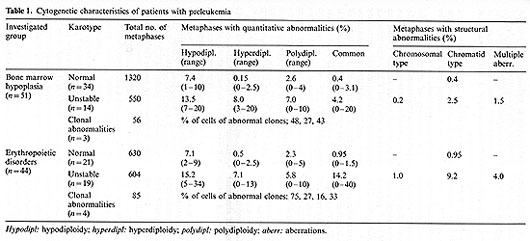
1. Mitelman F, Levan a (1981) Clustering aberrations to specific
chromosomes in human neoplasms. Hereditas 95:79-139 |